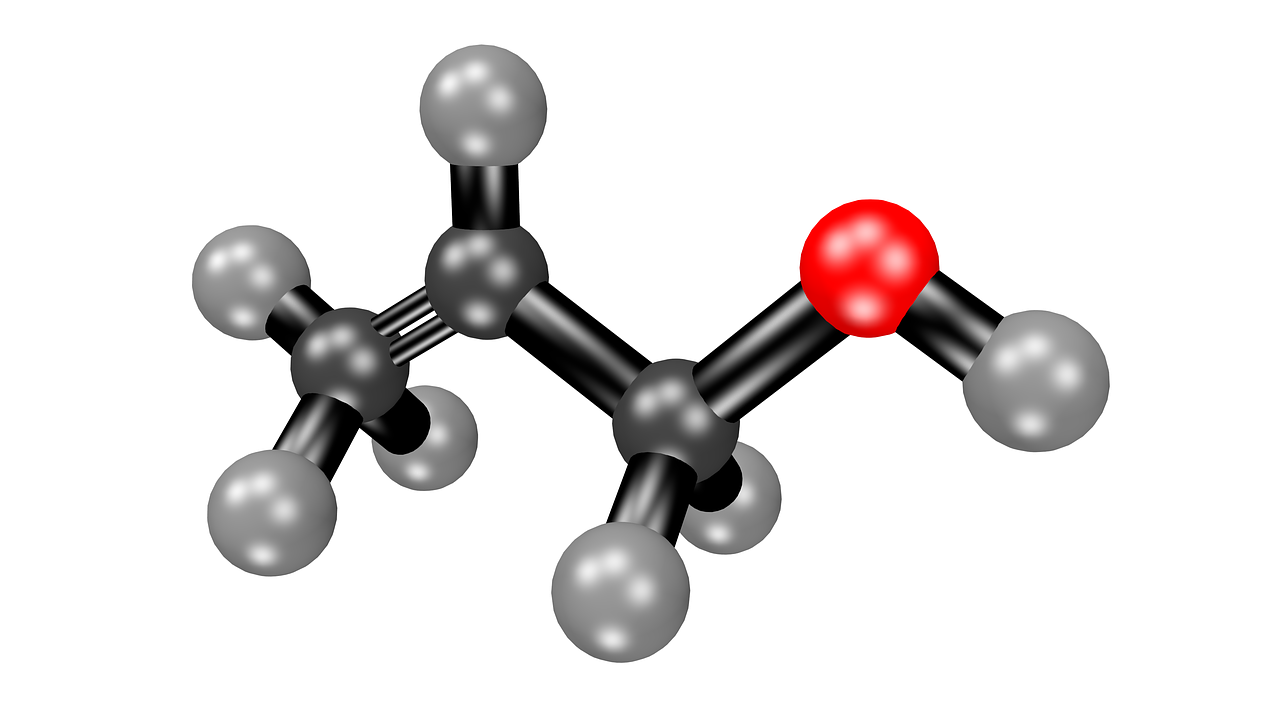
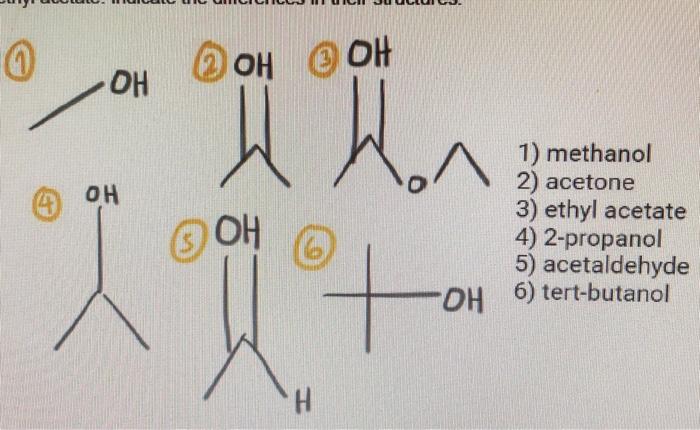
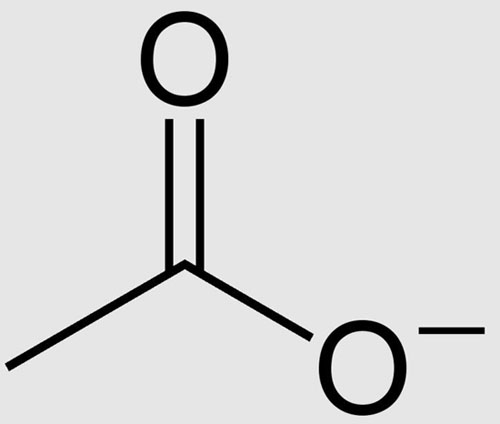
SOLVED: 1) A mixture of Ethyl acetate and water is used for extracting a compound. Discuss your observations and explain why? 2) There are several practices that need to be avoided during

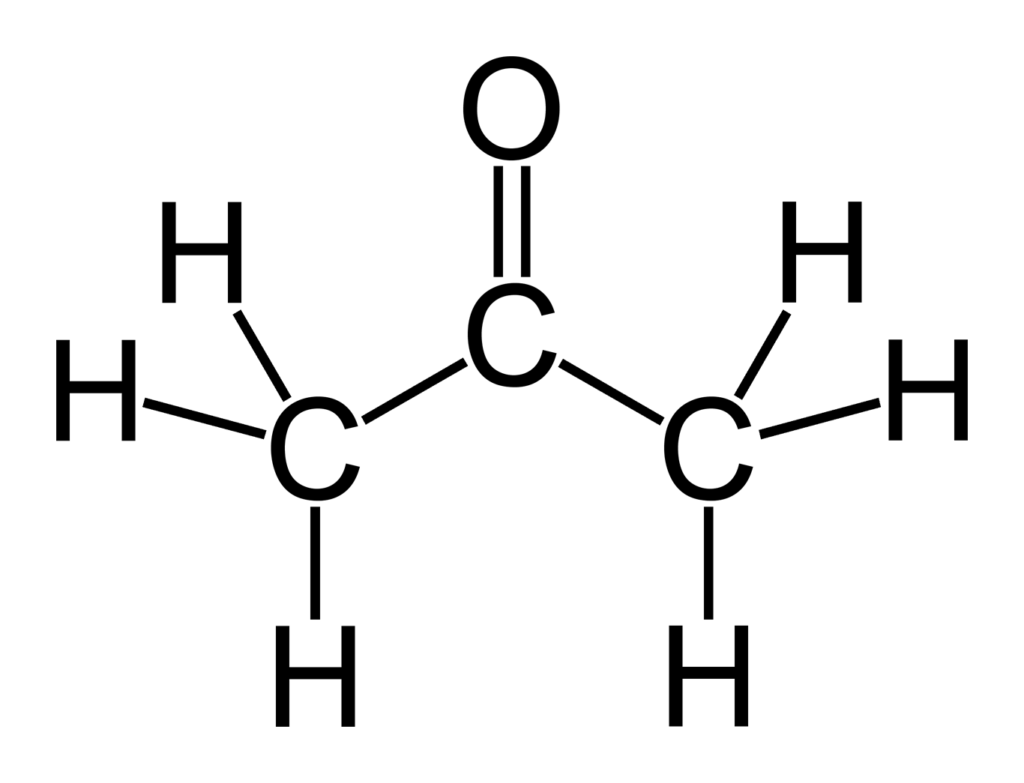
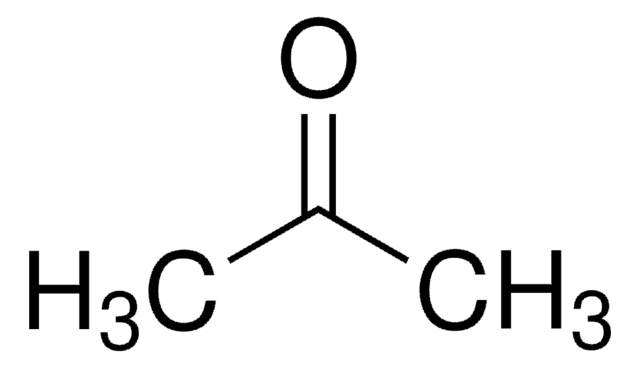
Write structures for acetone, a ketone, and methyl ethanoate, an ester. Based on your IR knowledge, compare the C=O bond lengths in these two compounds and discuss their placement on the IR
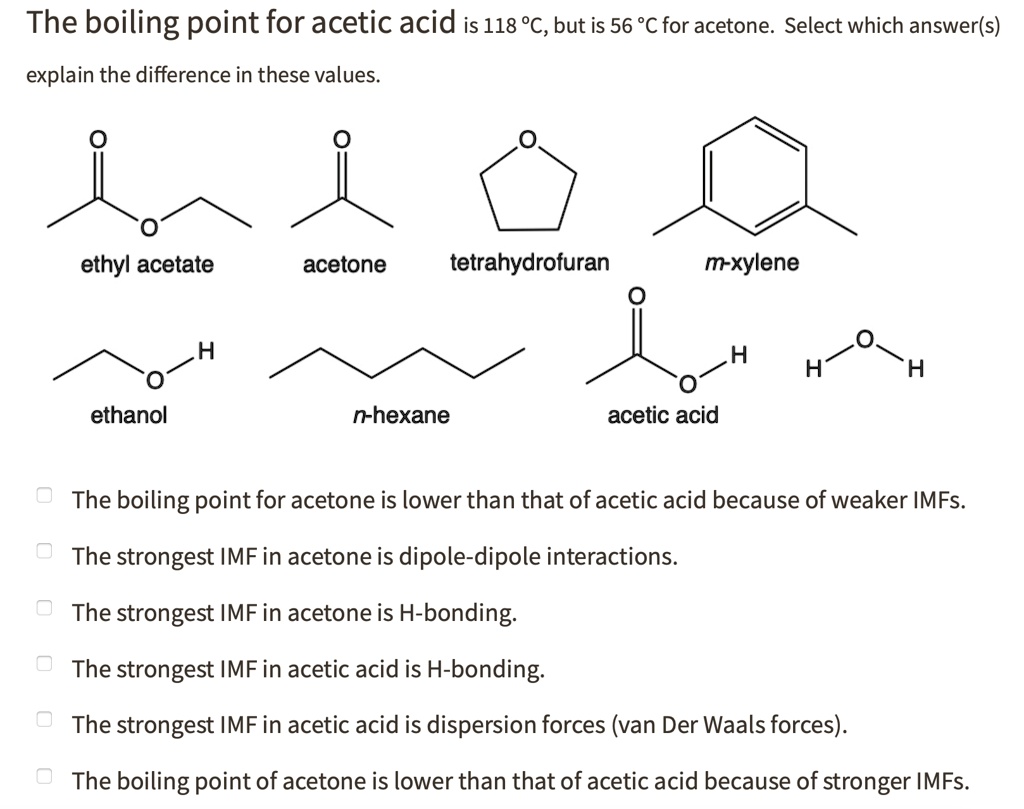
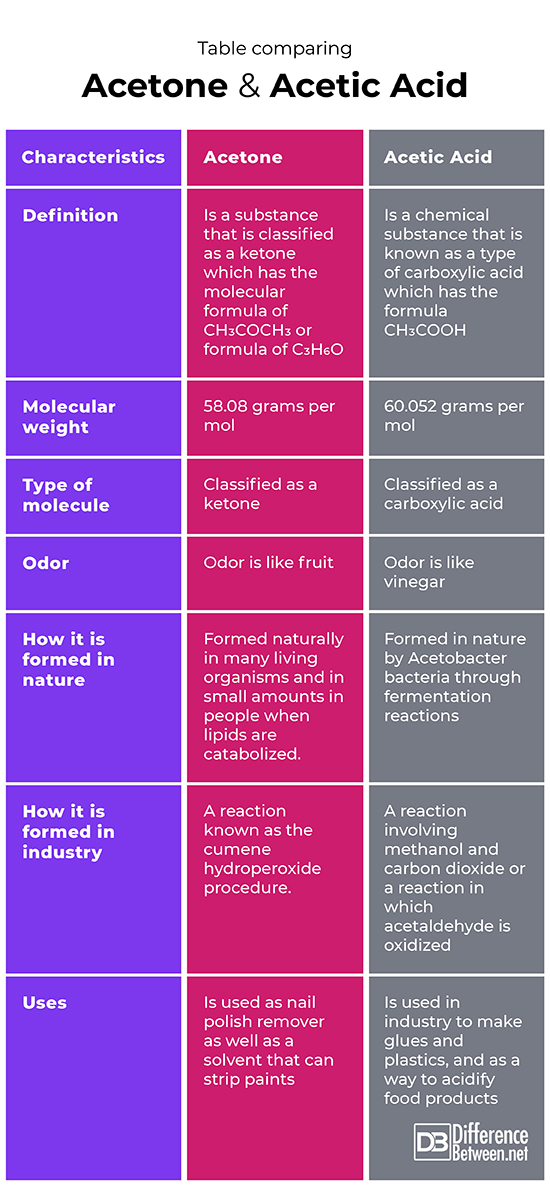
SOLVED: The boiling point for acetic acid is 118 'c, but is 56 'c for acetone. Select which answer(s) explain the difference in these values. ethyl acetate acetone tetrahydrofuran m-xylene ethanol n-hexane

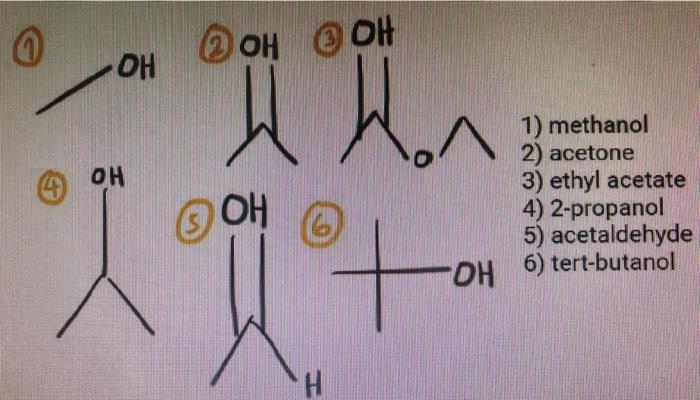
Arrange the following solvents in order of increasing polarity: a) ethanol b) ethyl acetate c) petroleum ether d) toluene e) acetone | Homework.Study.com